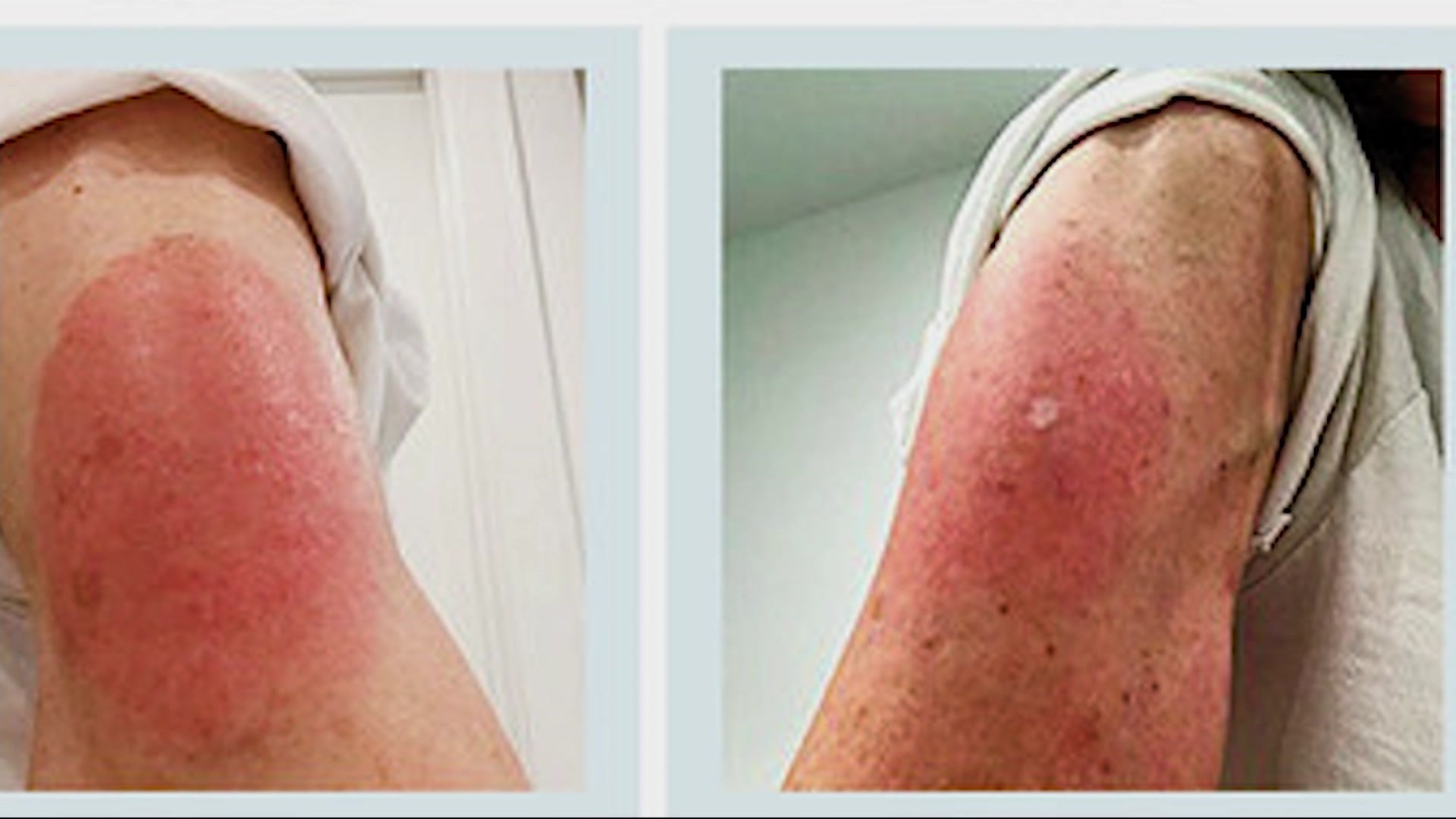
Each COVID-19 vaccine trial had to have at least 30,000 participants. There are bound to be more side effects observed and reported by the general population than are uncovered during a clinical trial, Perlman says. In rare cases, people have also developed shingles after COVID-19 vaccination, as small numbers of people do after taking other types of drugs, but those findings are preliminary. Others have experienced brain fog that feels akin to a marijuana high. A Moderna rep did not return requests for comment by press time.) There have also been reports of people developing rashes and other skin conditions, like so-called “ COVID arm,” after vaccination. A spokesperson for Janssen said menstrual changes were not tracked as part of its study. (In statements to TIME, a spokesperson for Pfizer said clinical trial participants did not report any menstrual changes. Menstrual changes may fall into that category, if conclusive evidence links them to the vaccines. In fact sheets describing each vaccine, manufacturers listed additional possible side effects-including diarrhea, joint pain, swollen lymph nodes, hives, rashes and facial swelling-and noted that there could be even more side effects beyond those specified. Centers for Disease Control and Prevention (CDC) lists common COVID-19 vaccine side effects as pain, swelling or redness at the injection site fatigue headaches muscle pain chills fever and nausea. Most side effects are unpleasant but normal, whereas serious adverse events are potentially fatal issues that demand medical care.) 
(Perlman says he considers side effects different from serious adverse events, like the rare blood clots reported in connection with Janssen/Johnson & Johnson’s shot. They show that the body is responding to the shot and mounting an immune response. Food and Drug Administration’s vaccine advisory committee. 
Stanley Perlman, a professor at the University of Iowa’s Carver College of Medicine and a member of the U.S. Experts on the advisory panel said the benefits of protecting children against COVID-19 outweigh the possible increased risk of the rare side effect.Side effects-while temporarily uncomfortable-are a standard part of vaccination, says Dr. The FDA recently authorized the use of Pfizer-BioNTech's COVID-19 vaccine in children ages 5 to 11 on an emergency basis. Most children and adolescents who experience myocarditis recover. However, viral infections, including COVID-19, also can cause myocarditis. The company estimated that more than 1.5 million adolescents had received its vaccine and said there didn't appear to be an increased risk of myocarditis in those younger than 18.īoth the Modern and Pfizer-BioNTech COVID-19 vaccines have been shown to produce myocarditis as a rare side effect. Moderna said it did not yet have access to the recent international analyses. The company's vaccine had previously been approved by The European Medicines Agency for use in children between ages 12 and 17. The FDA is reviewing "recent international analyses" of the possible side effect, according to Moderna.Ī recent unpublished study by Sweden's Public Health Agency showed a slightly increased risk of inflammation of the heart muscle and prompted four countries to stop giving the shot to young men and boys. Shots - Health News FDA authorizes use of Pfizer's COVID vaccine for 5- to 11-year-olds
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
